Introduction
Many of our chemistry and physics students experience research under the direction of the faculty. This research often leads to presentation at regional and national meetings typically during the summer months. Research also leads to publication in peer-reviewed journals. On the following pages you can find a summary of the research projects the faculty are currently involved in and a list of publications/presentations.
Exploring molecular and biochemical properties of the protein desmoplakin
Desmoplakin is a protein essential for cell-cell contacts. Point mutations in desmoplakin result in life threatening cardiomyopathies and skin diseases. Our goal is to learn about the molecular and biochemical properties of individual point mutations and how they affect the function of the native desmoplakin protein. With collaborations at other institutes around the country we will delve into the intricate structural biology of desmoplakin and its various mutations as well as use patient specific engineered tissue and mouse models to understand their physiology. Ultimately, we will use the knowledge we gain to develop novel therapeutics to treat life threatening diseases.
Coal mines, many of them shaft mines, are located in Tioga County in Northcentral Pennsylvania. Although abandoned over a century ago, the mines still heavily impact surrounding ecosystems by means of acid mine drainage (AMD) and the leaching of heavy metals into nearby streams and aquifers.
Two common iron (II) sulfide minerals, pyrite and marcasite, prevalent in mine tailings, are responsible for much of the AMD. When exposed to air, the following reaction occurs:
4FeS2 + 15O2 + 14H2O → 4Fe(OH)3 + 8H2SO4
Both iron and sulfur are oxidized in this reaction: iron (II) to iron (III), and the sulfide ion is oxidized to sulfate which further reacts to form sulfuric acid. These reactions are hazardous to the aquatic environment located at these sites. The iron (III) cation reacts with water easily to form iron (III) hydroxide. The sulfuric acid, being a very strong acid, dissociates quickly in water and lowers the pH of the environment to dangerous levels for the aquatic life present, as well as rendering the water unsuitable for many human uses.
My research focuses of several key areas. First, water samples from AMD impacted sources are measured using ICP-OES (Inductively Coupled Plasma – Optical Emission Spectrometry) to determine the concentration of many different heavy metals (iron, manganese, nickel, chromium, etc.) Second, aquatic plant (typically algae) samples from AMD-impacted locations are collected and processed through acid-digestion to measure the uptake of heavy metals by the plants. Third, samples (mushrooms) from nearby the AMD sources are collected and processed to look for uptake of heavy metal ions.
In the future, my research will expand to look at heavy metal ion concentrations in animals caught near AMD locations, and we will expand our search from just heavy metal ions to rare earth ions as well.
Project 1: Quantifying the accumulation and health effects of micro- and nanoplastic exposure in organismal models. The persistence of plastic polymers in the environment may pose serious health threats to a variety of organisms, as plastic debris is quite chemically durable. "Microplastics" are generally agreed upon to be 1μm-5mm; plastic particles smaller than 1μm are considered "nanoplastics". Both microplastics and nanoplastics are particularly worrisome as pollutants because their small sizes render them more bioavailable throughout the food chain. The potential health effects of plastic consumption must be systematically addressed with respect to dosage in a variety of organisms to determine the physical and chemical mechanisms of uptake and damage, and to guide ecological research regarding the manifestations of plastics at various trophic levels. The goals of this research are to (1) systematically investigate and quantify the effects of size and polymer composition on the health of plant, invertebrate, and vertebrate models; (2) to develop analytical protocols for quantifying both microplastic and nanoplastic content from tissues and environmental samples; and (3) to quantify the amount of under-sampling of nanoplastics from when protocols developed for microplastics are used. The organismal models currently being explored are mice (vertebrate), tardigrades (invertebrate), and different plants. This research project is collaboration between Dr. Farkas and Dr. Long (Mansfield Biology).
Project 2: Investigating cryptobiosis in tardigrades. Tardigrades are ubiquitous organisms found in almost every aquatic habitat on Earth. They are often thought of as "Schrodinger's organisms" for their ability to undergo cryptobiosis in response to external stresses; during cryptobiosis, tardigrades may be described as both living and dead due to an almost negligible metabolism! In response to slow dehydration, tardigrades express intrinsically disordered proteins which are thought to protect the structures of cells within. How protection is conferred, how it reverses, and the limits of tolerance for this response are not well quantified. In this project, the response of tardigrades to various osmotic stresses is being compared with that from slow dehydration, as osmobiosis is one of the least investigated mechanisms of cryptobiosis. This research project is collaboration between Dr. Farkas and Dr. Long (Mansfield Biology).
Improving yields of acceptor monomers in collaboration with Corning Inc.
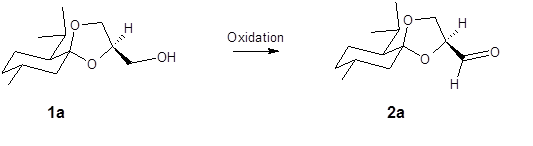
Oxidation of a glycerol menthonide isomer and its reactivity
Description of project:
Glycerol menthonide isomer 1a has been previously prepared but its chemistry has not been fully evaluated. The goal of this project will be the successful oxidation of 1a to yield the aldehyde, 2a. Confirmation of the product will be through various instrumentation such as but not limited to, NMR, FT-IR and GC/MS. Since 2a is a novel compound it will need to be fully documented for potential publication in a peer reviewed journal.